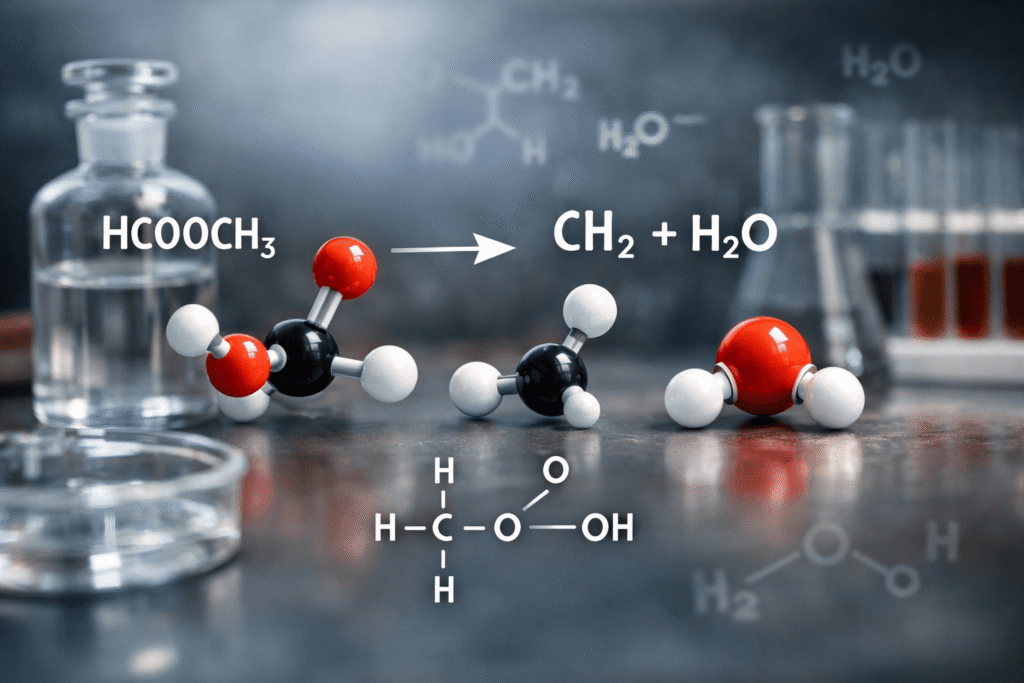
Have you ever wondered how a simple chemical reaction could power industries worth billions of dollars while sitting at the heart of your chemistry textbook? The notation HCOOCH CH2 H2O might appear cryptic at first glance, but it represents one of organic chemistry’s most fundamental and economically significant processes: the hydrolysis of methyl formate. This reaction doesn’t just break chemical bonds—it breaks through to real-world applications in pharmaceuticals, textiles, agriculture, and clean energy production.
Whether you’re a chemistry student preparing for exams, a researcher exploring ester cleavage mechanisms, or an industry professional optimizing production processes, understanding this reaction will transform how you perceive the molecular world around you. From the leather jacket in your closet to the preservatives keeping your food fresh, methyl formate hydrolysis touches your life in ways you’ve never imagined.
In this comprehensive guide, we’ll unravel the science behind HCOOCH CH2 H2O, explore both acid-catalyzed and base-catalyzed pathways, examine industrial applications, and reveal why this seemingly simple transformation remains crucial in modern chemistry. Let’s dive deep into the fascinating world of ester hydrolysis.
What Does HCOOCH CH2 H2O Actually Mean? Decoding the Chemical Notation
The expression “HCOOCH CH2 H2O” is an informal or shorthand representation of the methyl formate hydrolysis reaction. While it may look unusual, it describes the interaction between methyl formate (HCOOCH₃), the simplest ester in organic chemistry, and water (H₂O).
Understanding the Components
Methyl Formate (HCOOCH₃): This is an ester compound characterized by its clear, volatile nature and distinctive fruity aroma. As the simplest ester, it serves as an ideal model for studying nucleophilic acyl substitution mechanisms. The molecule contains a carbonyl group (C=O) bonded to an oxygen atom carrying a methyl group (OCH₃).
Water (H₂O): Far from being a passive solvent, water acts as the active nucleophile in this transformation. Its polar nature and ability to donate electron pairs make it perfect for attacking the electrophilic carbonyl carbon in the ester.
The CH2 Notation: While somewhat ambiguous in the original expression, this typically refers to the methylene group that, combined with an additional hydrogen from water, forms the methyl group (CH₃) in methyl formate.
The Correct Chemical Equation
The properly written hydrolysis reaction is:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This equation reveals the complete story: methyl formate reacts with water to produce formic acid (HCOOH) and methanol (CH₃OH)—two incredibly valuable industrial chemicals.
The Science of Ester Hydrolysis: Breaking Down the Reaction Mechanism

Ester hydrolysis represents a fundamental transformation in organic chemistry where ester bonds are cleaved by water molecules. This process is essentially the reverse of esterification, where acids and alcohols combine to form esters. Understanding both acid-catalyzed and base-catalyzed pathways is crucial for optimizing industrial processes.
Acid-Catalyzed Hydrolysis: The Reversible Pathway
In acidic conditions, the hydrolysis of methyl formate follows a specific mechanistic pathway:
Step 1 – Protonation: The carbonyl oxygen of methyl formate becomes protonated by the acid catalyst (typically H₂SO₄ or HCl). This protonation increases the electrophilicity of the carbonyl carbon, making it more susceptible to nucleophilic attack.
Step 2 – Nucleophilic Attack: A water molecule attacks the protonated carbonyl carbon, forming a tetrahedral intermediate. This intermediate is a crucial transitional structure containing both the original ester components and the attacking water molecule.
Step 3 – Proton Transfer and Bond Cleavage: Through a series of proton transfers, the intermediate breaks down. The ester bond cleaves, releasing methanol as the leaving group and forming formic acid.
Key Characteristic: This reaction is reversible, establishing an equilibrium between reactants and products. Industrial processes must remove products or use excess water to drive the reaction forward according to Le Chatelier’s principle.
Base-Catalyzed Hydrolysis: The Saponification Route
Base-catalyzed ester hydrolysis, also known as saponification, offers significant advantages in many applications:
Step 1 – Direct Nucleophilic Attack: The hydroxide ion (OH⁻) directly attacks the carbonyl carbon of methyl formate. The negative charge makes hydroxide a much stronger nucleophile than water.
Step 2 – Tetrahedral Intermediate Formation: The addition of the hydroxide creates an intermediate structure where the carbonyl carbon is temporarily bonded to four groups.
Step 3 – Elimination and Product Formation: The intermediate collapses, eliminating the methoxide ion (CH₃O⁻) and forming a formate anion. Upon acidification, this produces formic acid.
Critical Advantage: Unlike acid-catalyzed hydrolysis, the base-catalyzed pathway is irreversible. The formation of the stable carboxylate ion prevents the reverse reaction, making this method highly efficient for complete ester conversion—a principle extensively used in soap-making industries.
Factors Influencing Reaction Rate and Yield
Several critical variables control the efficiency of methyl formate hydrolysis:
Catalyst Type and Concentration: Strong acids like sulfuric acid or strong bases like sodium hydroxide dramatically accelerate reaction rates. Industrial processes carefully balance catalyst concentration with cost and safety considerations.
Temperature Effects: Research indicates that increasing temperature from 25°C to 60°C can boost hydrolysis rates by approximately 300%. However, excessive heat may trigger unwanted side reactions or decomposition.
Water Concentration: Using excess water shifts the equilibrium toward product formation. In industrial settings, this is balanced against processing costs and equipment capacity.
pH Control: The choice between acidic (pH < 7) or basic (ph > 7) conditions fundamentally alters the reaction pathway, affecting both speed and reversibility.
Industrial Applications: Where HCOOCH CH2 H2O Powers Real-World Processes
The hydrolysis of methyl formate isn’t just academic—it’s a cornerstone of modern industrial chemistry, generating products worth billions annually.
Formic Acid Production: A Global Industry
Formic acid (HCOOH), also known as methanoic acid, ranks among the most commercially significant products from this reaction. Global demand has exceeded 850 kilotons annually, driven by diverse applications:
Leather Processing: The tanning industry relies heavily on formic acid to enhance leather quality and durability. It helps regulate pH during the tanning process and improves the penetration of tanning agents.
Textile Industry: As a dyeing agent, formic acid improves color fastness and helps maintain consistent dye uptake across different fabric types.
Agricultural Preservatives: Farmers use formic acid as a silage preservative to prevent harmful bacterial growth in stored animal feed. This application protects livestock health and reduces feed waste.
Chemical Synthesis: Formic acid serves as a reducing agent in various chemical processes and as a feedstock for producing other valuable chemicals.
Green Energy Applications: Emerging research explores formic acid as a hydrogen carrier for fuel cell technologies, positioning it at the forefront of clean energy innovation.
Methanol: The Versatile Industrial Workhorse
Methanol (CH₃OH), the alcohol product of methyl formate hydrolysis, demonstrates remarkable versatility:
Fuel Production: Methanol serves as an alternative fuel source and antifreeze agent. Its potential in fuel cells highlights its growing importance in sustainable energy systems.
Chemical Feedstock: Industries use methanol to produce formaldehyde, acetic acid, and various plastics. It’s a fundamental building block for countless synthetic materials.
Solvent Applications: The paint, pharmaceutical, and adhesive industries extensively employ methanol as a solvent due to its excellent dissolving properties.
Biodiesel Production: Methanol plays a crucial role in transesterification reactions that convert vegetable oils into biodiesel fuel.
Pharmaceutical and Fragrance Industries
The principles of ester hydrolysis extend beyond methyl formate to numerous pharmaceutical and fragrance applications. Understanding hydrolysis mechanisms helps chemists:
- Predict the stability and shelf life of ester-basedmedications
- Design fragrance compounds with desired volatility and longevity
- Develop controlled-release drug formulations
- Create synthetic flavoring agents for food products
Educational Significance: Why Students Must Master This Reaction
For chemistry students, methyl formate hydrolysis serves as more than just another equation to memorize—it’s a gateway to understanding fundamental organic chemistry principles.
Core Concepts Illustrated
Nucleophilic Substitution Mechanisms: The reaction perfectly demonstrates how nucleophiles attack electrophilic centers, a pattern repeated throughout organic chemistry.
Acid-Base Catalysis: Students learn how catalysts accelerate reactions without being consumed, a principle essential for industrial chemistry.
Chemical Equilibrium: The reversibility of acid-catalyzed hydrolysis provides concrete examples of Le Chatelier’s principle in action.
Reaction Kinetics: Studying how temperature, concentration, and catalysts affect reaction rates builds intuition for optimizing chemical processes.
Common Exam Topics and Practice Problems
Chemistry educators frequently test students on ester hydrolysis through various question types:
Mechanism Drawing: Students must illustrate arrow-pushing mechanisms showing electron movement during each reaction step.
Product Prediction: Given different esters and conditions, students identify the carboxylic acid and alcohol products.
Comparison Questions: Contrasting acid-catalyzed versus base-catalyzed pathways tests deeper understanding of reaction mechanisms.
Industrial Application Scenarios: Real-world problems require students to select optimal conditions for specific manufacturing goals.
Practice Problem Example
Question: Predict the products when HCOOCH₃ reacts with excess hot, dilute NaOH.
Answer: This represents base-catalyzed hydrolysis (saponification). The products are the sodium formate salt (HCOONa) and methanol (CH₃OH). Upon acidification with HCl, sodium formate converts to formic acid (HCOOH).
Comparing Related Ester Hydrolyses: Understanding the Differences
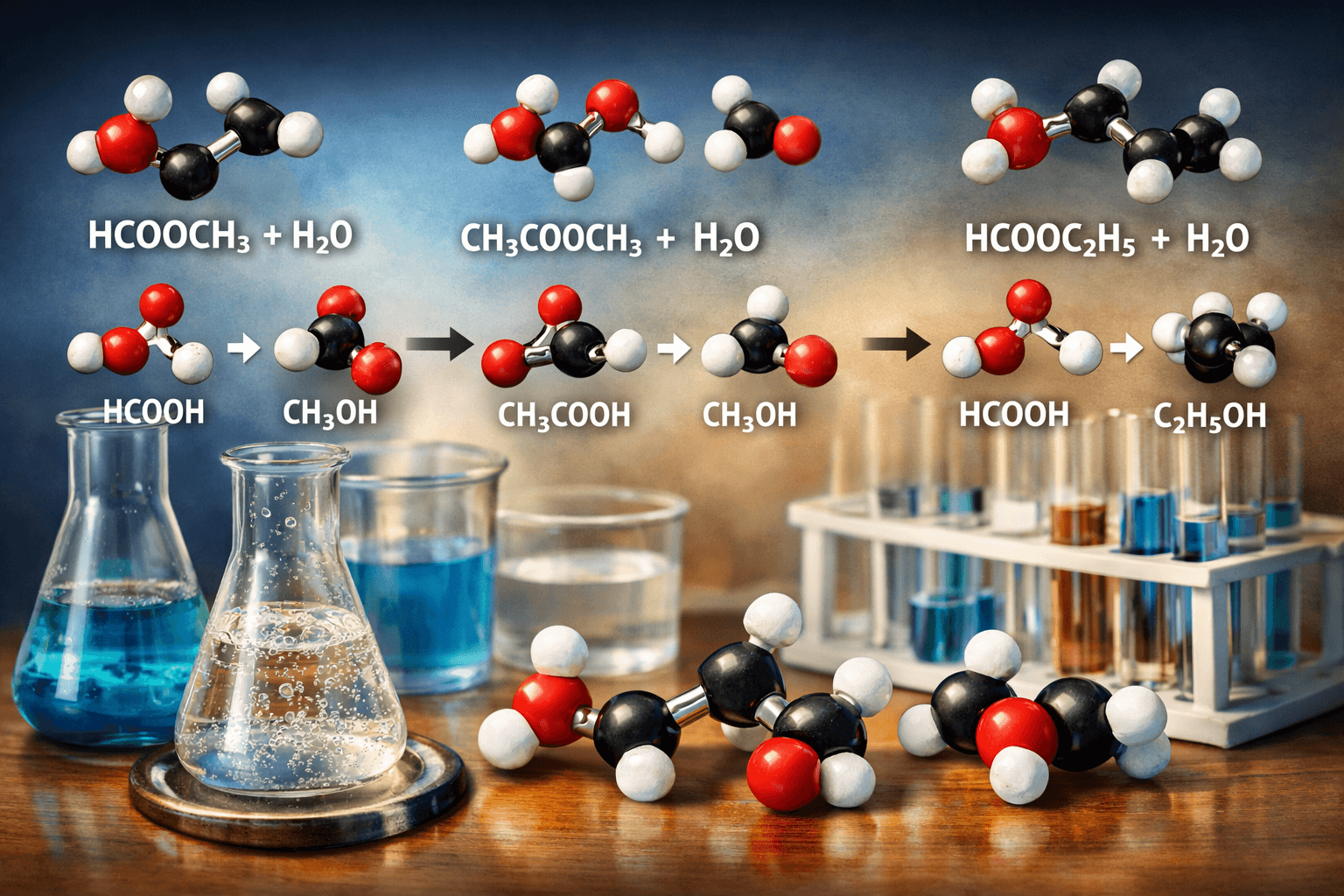
While methyl formate serves as an excellent model compound, understanding how different esters behave during hydrolysis provides valuable insights.
Methyl Formate vs. Methyl Acetate
Methyl Acetate Hydrolysis: CH₃COOCH₃ + H₂O → CH₃COOH + CH₃OH
This reaction produces acetic acid instead of formic acid. While mechanistically similar, the additional methyl group in methyl acetate slightly alters reaction kinetics and product properties.
Methyl Formate vs. Ethyl Formate
Ethyl Formate Hydrolysis: HCOOCH₂CH₃ + H₂O → HCOOH + CH₃CH₂OH
Here, ethanol forms instead of methanol. The larger alkyl group affects the ester’s volatility and solubility, influencing industrial processing conditions.
Key Distinguishing Factors
Volatility: Methyl formate evaporates more readily than larger esters, affecting storage and handling procedures.
Toxicity: Methanol produced from methyl formate requires more careful handling than ethanol from ethyl formate due to its higher toxicity.
Industrial Preference: Methyl formate hydrolysis is often preferred for formic acid production due to higher efficiency and simpler purification processes.
Safety Considerations and Environmental Impact
Working with HCOOCH CH2 H2O reactants and products requires understanding potential hazards and implementing proper safety protocols.
Handling Methyl Formate
Flammability: Methyl formate is highly flammable with a low flash point. Storage requires proper ventilation and flame-free environments.
Inhalation Risks: Vapors can cause respiratory irritation. Work areas must have adequate ventilation systems.
Personal Protective Equipment: Safety goggles, gloves, and lab coats are essential when handling methyl formate.
Formic Acid Hazards
Corrosivity: Formic acid can cause severe skin burns and eye damage. Immediate washing is crucial following accidental contact.
Vapor Exposure: Inhalation of formic acid vapors irritates respiratory passages. Fume hoods are mandatory for reactions generating these vapors.
Methanol Toxicity
Ingestion Dangers: Methanol is highly toxic when ingested, potentially causing blindness or death. Never consume or allow accidental ingestion.
Dermal Absorption: Skin contact allows methanol absorption, requiring prompt washing with soap and water.
Proper Disposal: Never pour methanol down drains. Follow institutional chemical waste disposal protocols.
Environmental Considerations
Modern industrial processes emphasize sustainability:
Green Chemistry Approaches: Researchers develop heterogeneous catalysts that reduce waste and allow easier catalyst recovery and reuse.
Biodegradability: Both formic acid and methanol biodegrade relatively quickly, though proper waste treatment remains essential.
Energy Efficiency: Optimizing reaction temperatures and catalyst concentrations reduces energy consumption in industrial ester hydrolysis processes.
Advanced Topics: Current Research and Future Directions
The chemistry of methyl formate hydrolysis continues evolving as researchers pursue more efficient and sustainable processes.
Continuous Flow Reactors
Modern chemical engineering employs continuous-flow millireactors for methyl formate hydrolysis, offering several advantages:
- Better temperature control for consistent product quality
- Reduced reaction times through optimized mixing
- Enhanced safety from smaller reaction volumes
- Easier scale-up from laboratory to industrial production
Research using acidic ion-exchange resins like Amberlyst 15 as catalysts shows substantially higher yields when feed compositions exceed methyl formate saturation in water.
Green Chemistry Innovations
Enzymatic Catalysis: While most industrial processes use chemical catalysts, research explores lipase-catalyzed ester reactions for more environmentally friendly processing.
Alternative Solvents: Scientists investigate ionic liquids and other green solvents to replace traditional organic solvents in ester synthesis and hydrolysis.
Waste Valorization: Converting waste syngas from steel manufacturing to formic acid and methanol through methyl formate intermediates exemplifies circular economy principles.
Computational Chemistry Applications
Modern research employs computational modeling to:
- Predict optimal catalyst structures for improved ester hydrolysis rates
- Understand solvent effects on reaction mechanisms
- Design novel catalysts with enhanced selectivity and activity
- Model industrial reactor performance before physical construction
Common Misconceptions and Clarifications
Myth 1: “Hydrolysis Always Requires Extreme Conditions”
Reality: While heating accelerates ester hydrolysis, the reaction occurs slowly even at room temperature. This fact becomes important when considering ester shelf life and storage stability.
Myth 2: “All Esters Hydrolyze Identically”
Reality: Ester structure significantly affects hydrolysis rates. Tertiary alkyl esters may follow SN1 mechanisms rather than the typical addition-elimination pathway, while primary esters consistently follow nucleophilic addition mechanisms.
Myth 3: “More Catalyst Always Means Faster Reaction”
Reality: Beyond optimal concentrations, additional catalyst provides diminishing returns and may actually promote unwanted side reactions or equipment corrosion.
Practical Applications in Laboratory Settings
For students and researchers conducting ester hydrolysis experiments, following best practices ensures success:
Setting Up the Reaction
- Measure reactants precisely using calibrated equipment
- Add catalyst slowly while monitoring temperature
- Maintain stirring to ensure homogeneous mixing
- Control temperature within optimal ranges (typically 60-80°C for faster kinetics)
- Monitor reaction progress through titration or spectroscopic methods
Product Isolation and Purification
Distillation: Separate methanol and formic acid based on their different boiling points
Extraction: Use liquid-liquid extraction to separate products from catalyst residues
Neutralization: In base-catalyzed reactions, acidification converts formate salts to formic acid
Drying: Remove residual water from products using appropriate drying agents
Frequently Asked Questions
Q1: Is HCOOCH CH2 H2O a single chemical compound?
No, it’s shorthand notation describing a reaction mixture containing methyl formate (ester), water, and the reaction products formic acid and methanol.
Q2: What are the final products of the HCOOCH CH2 H2O reaction?
The hydrolysis produces two main products: formic acid (HCOOH) and methanol (CH₃OH).
Q3: Is this reaction reversible?
Acid-catalyzed hydrolysis is reversible, establishing equilibrium. Base-catalyzed hydrolysis (saponification) is effectively irreversible due to stable carboxylate ion formation.
Q4: Why is this reaction important in real life?
It’s a major industrial route for producing formic acid and methanol, chemicals essential in leather processing, textiles, agriculture, pharmaceuticals, and clean energy applications.
Q5: Can I perform this reaction at home?
While theoretically possible, it’s not recommended due to safety hazards. Methyl formate is flammable, formic acid is corrosive, and methanol is toxic. Laboratory facilities with proper safety equipment are necessary.
Q6: How does temperature affect the reaction?
Higher temperatures dramatically increase reaction rates—studies show approximately 300% rate increases when heating from 25°C to 60°C. However, excessive heat may cause decomposition or unwanted side reactions.
Q7: What’s the difference between acid and base catalysis for this reaction?
Acid catalysis creates a reversible equilibrium, while base catalysis produces irreversible saponification. Acid methods often provide simpler product isolation, while base methods ensure complete conversion.
Conclusion
The seemingly simple notation HCOOCH CH2 H2O opens doors to understanding one of organic chemistry’s most fundamental and industrially significant processes. From its role in producing essential chemicals like formic acid and methanol to its educational value in teaching nucleophilic substitution mechanisms, methyl formate hydrolysis exemplifies how basic chemical principles drive real-world applications.
Whether you’re a student mastering ester cleavage for exams, a researcher optimizing industrial processes, or simply curious about the chemistry shaping modern life, understanding this reaction provides invaluable insights.
As industries push toward greener, more sustainable chemical production, reactions like methyl formate hydrolysis will continue evolving, incorporating new catalysts, reactor designs, and processing conditions.
The next time you encounter leather products, preserved foods, or discussions about clean energy, remember the molecular transformation represented by HCOOCH CH2 H2O—a testament to how simple chemical reactions power complex industrial ecosystems and improve daily life across the globe.